Research in the Mani Group focuses on photo- and radiation- induced fundamental chemical reactions in condensed phase. We are investigating and developing molecules with quantum functionalities. The goal of our research program is to understand how to control electronic excited states, charge and exciton transfer reactions, and spin dynamics in these molecules and molecular assemblies. The group is also interested in exploiting new knowledge gained to pursue developments of new quantum technologies from biomedical imaging to energy applications. Our research approach is a seamless transition between synthetic chemistry and physical chemistry. We combine molecular design/synthesis with computational chemistry and spectroscopic techniques including ultrafast laser spectroscopy (visible to IR) and pulse radiolysis.
Active Projects
Spin-Correlated Radical Pairs and the Associated Emissions for Quantum Information Science
Spin-correlated radical pairs (SCRPs) can act as spin qubit pairs (SQPs). We are exploring the possibility of using SCRPs of organic molecules in the development of new quantum information technologies with special focus on quantum sensing (Chemical Physics Review 2022).
Long-range electron transfer processes are not uncommon in natural and artificial systems. The subsequent long-range radical pairs or charge-separated states typically recombine into the electronic ground state non-radiatively (without emission). Yet, certain molecular systems can exhibit radiative charge recombination from these radical pairs. This is generally referred to as charge-transfer (CT) emission. While short-range CT emissions are commonly observed, long-range CT emission (beyond contact distance) is very rare. We are exploring the ways to realize efficient CT emission over long distance, intramolecular long-range CT emission. Such emission can provide unique opportunities to control molecular emission. Recently, we have designed the molecular systems that give CT emission at distances to 24 Å operate at room temperature, presenting a generally applicable design principle of intramolecular long-range CT emission (JPC Letters, 2019). We also observed an efficient delayed fluorescence (charge recombination fluorescence) from the initial singlet excited states that are re-generated from another charge recombination of the long-range radical pairs. We have demonstrated the magnetic control of recombination fluorescence and tunability of field response range (JACS, 2020). Further developments of SCRPs-based quantum control and molecular qubits (e.g., JACS, 2025) are currently ongoing!
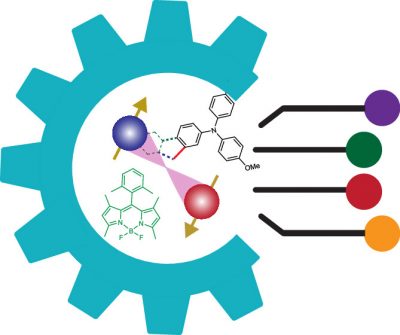
Use of Vibrational Probes to Understand Charges and CS States
We employ vibrational spectroscopy to unravel the nature of charges and CS states. In particular, we take advantage of and push the limits of the sensitivity of nitrile (CN) vibrations. We have shown that the nitrile vibration acts as an excellent vibrational probe for radical anions. They are sensitive to the induced electric field and the delocalization of electrons (JACS 2015#1, JACS 2015 #2, JPC-B 2016). Our recent works show that we can gain unique insights into the energy and dynamics of free charges (JPC-B 2017 , JPC-B 2020) as well as those of CS states (JPC-A 2018). We showed that the IR linewidth and intensities, in addition to the frequency shifts, can provide complimentary and unique information (Chem Sci 2021).

Heavy-Atom-Free Photoredox Catalysis using Singlet and Triplet Excited States of Organic Molecules
Triplet excited states are important electronic states, but those of organic molecules are underutilized. We explore ways to efficiently produce triplet excited states of organic molecules without heavy atoms such as precious metals, and utilize them in various applications including organic synthesis, molecular electronics, and energy technologies.
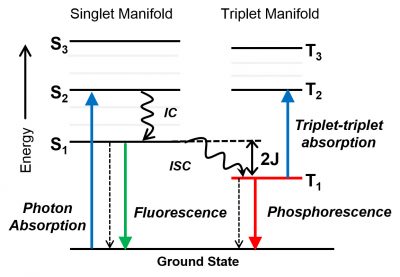 One of the mechanisms we are actively pursuing is spin-orbit, charge-transfer intersystem crossing (SOCT-ISC). SOCT-ISC is one of the promising mechanisms that can produce triplet excited states of organic molecules without using heavy-atom effects. We have recently uncovered the energetic requirements for this mechanism, establishing one of the general design criteria for donor-acceptor molecules that can produce triplet excited states efficiently (Chem, 2019). We used this new class of molecules in triplet-triplet upconversion under aerobic conditions using a unique soft mater system (ACS AMI, 2020) as well as a photocatalyst in organic synthesis (ACS Omega, 2021).
One of the mechanisms we are actively pursuing is spin-orbit, charge-transfer intersystem crossing (SOCT-ISC). SOCT-ISC is one of the promising mechanisms that can produce triplet excited states of organic molecules without using heavy-atom effects. We have recently uncovered the energetic requirements for this mechanism, establishing one of the general design criteria for donor-acceptor molecules that can produce triplet excited states efficiently (Chem, 2019). We used this new class of molecules in triplet-triplet upconversion under aerobic conditions using a unique soft mater system (ACS AMI, 2020) as well as a photocatalyst in organic synthesis (ACS Omega, 2021).